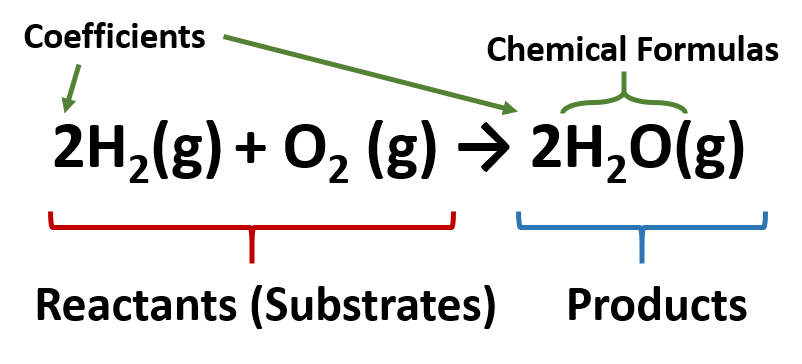
A chemical reaction represents a change in the distribution of atoms but not in the number of atoms. A balanced chemical equation is when both the numbers of each type of atom and the total charge are the same on both sides. Abbreviations are added in parentheses as subscripts to indicate the physical state of each species: - ( s ) for solid, ( l ) for liquid, ( g ) for gas, and ( aq ) for an aqueous solution. An arrow, read as yields or reacts to form, points from the reactants to the products. A chemical equation is an expression that gives the identities and quantities of the substances in a chemical reaction Chemical formulas and other symbols are used to indicate the starting material(s) or reactant(s), which are written on the left side of the equation, and the final compound(s) or product(s), which are written on the right side.Can a redox reaction form a molecule? Explain your answer. What is the sum of the oxidation numbers in a compound? 31. What is the oxidation number of the fluorine ion? 24. What is the oxidation number of an element? 23. What is the general form of a double displacement reaction? 21. What determines whether one metal will replace another in a single displacement reaction? 20. What type of reaction is an explosion? 18. How is a combustion reaction related to a synthesis reaction? 17. Balance the equation for the reaction of magnesium chloride and silver nitrate to form magnesium nitrate and silver chloride. What is the procedure for balancing a chemical equation? 14. What the three things does conservation of matter require of chemical equations? 13. How do you indicate a catalyst is being used in a reaction? 11. What symbols are used to represent the states of matter? 9.

What information is found in an equation? 8. What is evidence a reaction has occurred? 3. Chemical Reaction - Study Questions 1.Multiply the two half-reactions by the number that yields the lowest common number of electrons in each half-reaction.The second half-reaction has 2- on the left and 0 on the right. Add 5 electrons to the left to balance the charge.

In this example, the first half-reaction has a charge of 7+ on the left and 2+ on the right.
Write a skeleton ionic equation that only covers the atoms that change oxidation number: MnO 4 - → Mn 2+ I - → I 2.Find the atoms that experience a change in oxidation number: Mn: +7 → +2 I: +1 → 0.Write down the oxidation numbers for each type of atom on both sides of the equation: Left hand side: K = +1 Mn = +7 O = -2 I = 0 H = +1 S = +6 Right hand side: I = 0 Mn = +2, S = +6 O = -2.First, write the unbalanced chemical equation: KMnO 4 + KI + H2SO 4 → I 2 + MnSO 4.
